Understanding what ppm stands for in gas analysis can save lives, prevent costly errors, and ensure industrial compliance.
The term ppm (parts per million) is a crucial unit of measurement used in gas analyzers to quantify the concentration of gases in a sample. Its accuracy is essential for safety, efficiency, and regulatory compliance across industrial applications.

What does ppm mean in gas analysis?
When you see ppm on a gas analyzer, you’re looking at the unit used to express gas concentration — parts per million.

In gas detection, 1 ppm means one part of a specific gas in one million parts of air. For example, 50 ppm of CO means there are 50 molecules of carbon monoxide per one million air molecules. This unit is essential because even trace amounts of certain gases can be hazardous or affect product quality.
Modern gas analyzers use sensors that detect changes in chemical or physical properties when exposed to gas samples.
Different technologies such as infrared absorption, electrochemical sensing, or thermal conductivity allow analyzers to quantify gases precisely in ppm. These values are then processed through digital algorithms to provide real-time readings. Sensor calibration and sensitivity directly affect the reliability of the ppm measurement.
Why is accurate measurement essential for industrial safety?
Ignoring or misreading ppm levels can lead to safety hazards, including toxic exposure or combustion risks.
In industries like oil & gas or chemical processing, exceeding safe ppm thresholds for gases like H₂S or CO can be life-threatening. An accurate ppm reading triggers alarms and system shutdowns before dangerous levels are reached, making it a frontline defense in gas leak detection.
Portable analyzers may measure ppm levels in a narrow range for quick diagnostics, while fixed analyzers offer continuous, high-precision monitoring. Some analyzers are designed for trace-level ppm detection, such as in toxic gas monitoring, while others handle broader ranges used in combustion control.
How do you calibrate a gas analyzer for accurate ppm readings?
Regular calibration ensures your analyzer continues to provide accurate ppm data under varying conditions.
Calibration involves exposing the analyzer to a known gas concentration — a gas calibration standard — to adjust its response accordingly. Depending on the application, calibration should be done monthly, weekly, or even daily, especially in environments with temperature, humidity, or pressure fluctuation
How do you calibrate a gas analyzer for accurate ppm readings?
Even the most advanced gas analyzer becomes unreliable without proper calibration — especially when precise ppm readings are required.
Calibration is the process of setting or correcting the gas analyzer to ensure its ppm readings are accurate when compared to a known reference value. It ensures that your analyzer interprets gas concentrations correctly, maintaining measurement integrity across changing environmental or operational conditions.
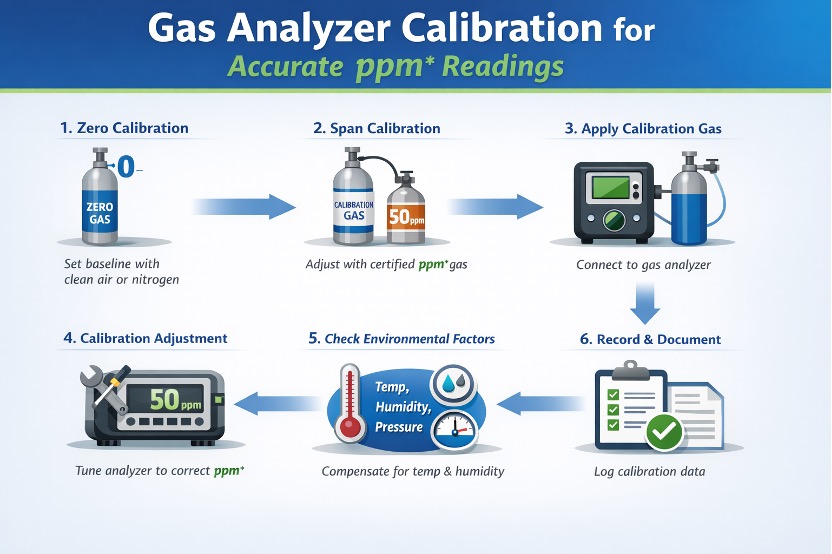
1. Why Calibration Matters in ppm Measurement
Small errors in ppm can have big consequences — particularly when measuring toxic or explosive gases. For instance, a miscalibration of 10 ppm in hydrogen sulfide (H₂S) detection can mean the difference between a safe environment and one that violates occupational exposure limits. Gas leak detection systems that rely on uncalibrated analyzers might trigger false alarms or, worse, fail to detect real threats.
2. Types of Calibration: Zero and Span
- Zero calibration involves exposing the analyzer to a gas with zero concentration of the target component (often clean, dry air or nitrogen). This sets the baseline response of the sensor.
- Span calibration uses a gas calibration standard with a precisely known ppm concentration of the target gas. This ensures the analyzer reads accurately across its measurement range.
Both steps are critical, especially for analyzers operating in dynamic environments such as refineries, chemical plants, or power stations.
3. Using Certified Gas Calibration Standards
Calibration gases must be traceable to national or international standards, such as NIST. The cylinder should clearly state the gas type, concentration in ppm, and uncertainty level. Using uncertified gases can lead to calibration drift, unreliable readings, and ultimately, regulatory non-compliance.
4. Recommended Calibration Frequency
- Daily or weekly for analyzers in harsh environments or safety-critical applications (e.g., toxic gas monitoring)
- Monthly or quarterly for general industrial use
- Always after sensor replacement, relocation, or exposure to extreme conditions
Many modern gas analyzers have auto-calibration features or prompt operators when calibration is due, helping maintain consistent ppm accuracy.
5. Environmental Conditions and Calibration Accuracy
Temperature, humidity, and atmospheric pressure can affect sensor response. It’s important to calibrate the analyzer under conditions similar to its typical operating environment, or use compensation algorithms if the environment is highly variable.
6. Calibration Documentation and Compliance
Proper documentation of calibration records — including date, technician name, gas type, and ppm value — is often legally required. Industries under OSHA, EPA, or ISO standards must be audit-ready with calibration logs to prove their ppm data is valid and trustworthy.
Which industries rely most on precise ppm detection?
Industries dealing with combustible, toxic, or reactive gases require highly sensitive monitoring.

Applications include environmental compliance in emissions monitoring, leak detection in LNG plants, and purity control in semiconductor manufacturing. Even slight deviations in ppm can result in fines, shutdowns, or compromised product integrity.























