Many engineers still think gas sampling system is simple. Just extract gas from the pipeline and send it to the gas analyzer. However, industrial gas analysis does not work that way. In real applications, the sampling system often determines whether the gas analyzer data is accurate or completely misleading. In fact, many “analyzer problems” actually begin long before the gas reaches the analyzer itself.
From the process duct to the gas analyzer cabinet, sample gas usually travels through multiple stages, including extraction, filtration, heating, transport, and conditioning. During this process, even a small design mistake can change the gas composition and distort the final measurement.
As a result, condensation, adsorption, corrosion, and particulate contamination become common hidden problems in industrial gas analysis systems. Once these issues appear, measurement drift, unstable readings, and instrument damage usually follow.
That is why experienced engineers no longer describe gas sampling as simple “gas extraction.” Instead, they treat it as a controlled process that protects the original condition of the sample gas from beginning to end. Simply put, if the gas changes before analysis, the gas analyzer can never show the real process condition.
What Makes a Gas Sampling System Truly Reliable?
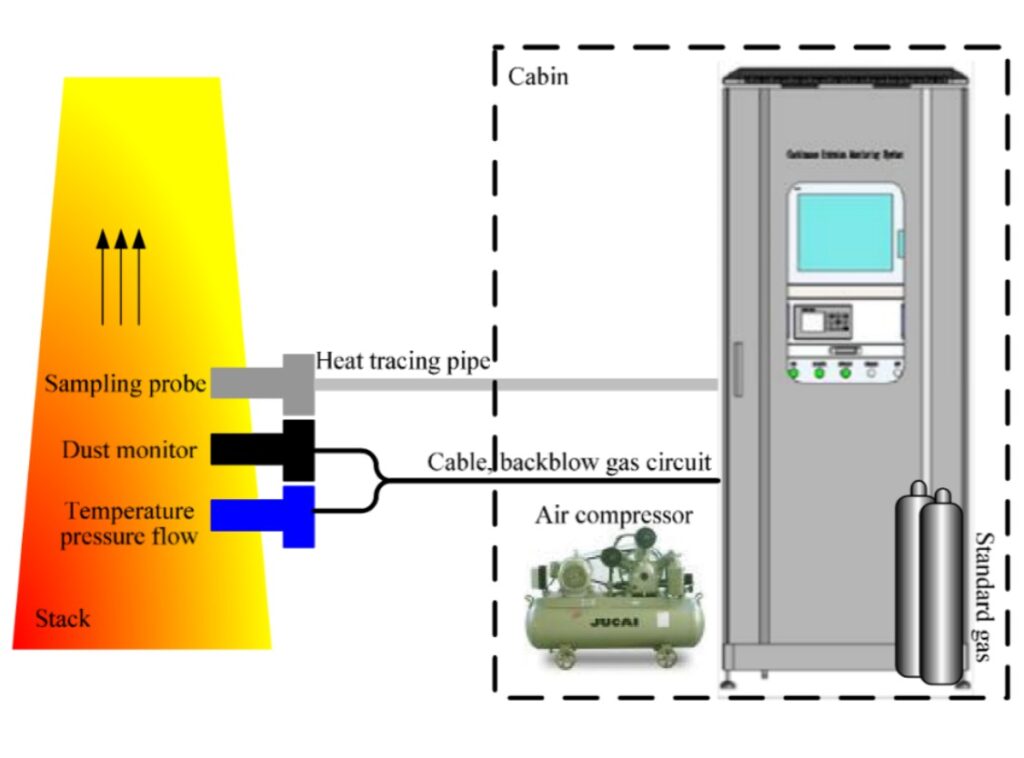
A reliable gas sampling system follows one core principle: Keep the sample representative, stable, and unchanged from extraction to analysis. To achieve that goal, four conditions must always be satisfied.
1. Why Must the Sample Be Representative?
The sample must accurately reflect actual process conditions. That sounds obvious, yet many systems fail here first. Probe location, gas flow distribution, and duct turbulence all influence sample quality. If the probe sits in a dead zone or near the duct wall, the measured composition may differ greatly from the real process condition. Therefore, proper probe positioning is critical from the beginning.
2. Why Must Gas Composition Remain Intact During Transport?
Once extracted, the gas should remain chemically and physically unchanged. Unfortunately, this is exactly where many systems lose accuracy. During transport, gases may condense, adsorb onto tubing surfaces, or react with contaminants. Even small changes create major measurement errors. For this reason, heated transport and inert materials become essential. PTFE-lined tubing, corrosion-resistant probes, and stable temperature control all help preserve sample integrity.
3. Why Must the System Handle Harsh Industrial Conditions?
Industrial gas analysis rarely happens under ideal conditions. Power plants, steel mills, cement kilns, and waste incinerators often operate with high dust concentration, acidic gases, and extreme moisture levels. Under these conditions, ordinary components fail quickly.
As a result, industrial sampling systems require:
- Corrosion-resistant materials
- High-temperature probes
- Multi-stage filtration
- Reliable heat tracing
- Anti-clogging structures
Without these protections, maintenance frequency increases dramatically.
4. Why Is Long-Term Stability So Important?
Accurate data is meaningless if the system cannot operate continuously. Industrial facilities rely on stable online monitoring for process control, environmental compliance, and safety management. Frequent downtime increases operational risk and maintenance cost. That is why modern sampling systems include temperature alarms, flow monitoring, and automatic back-purge functions to maintain stable operation over long periods.
What Are the 4 Hidden Problems That Most Gas Sampling Systems Fail to Prevent?

Hidden Problem #1: Why Does Condensation Cause Major Measurement Errors?
Condensation is one of the most overlooked problems in gas sampling systems. At first glance, a small amount of moisture inside the tubing may not seem serious. However, the impact on gas analyzer accuracy can be enormous.
When hot sample gas cools below its dew point, water vapor condenses into liquid droplets. During this phase change, many target gases partially dissolve into the condensate. As a result, the gas analyzer receives a chemically altered sample instead of the original process gas. This issue becomes especially serious in flue gas applications containing acidic components such as SO₂ and NOx. Once acidic condensate forms, corrosion and sensor degradation accelerate rapidly. To prevent this problem, industrial systems typically use heated sample lines and heated probes. Their purpose is straightforward: maintain the entire sample path above the acid dew point. In other words, stable temperature control is not optional. It is essential.
Hidden Problem #2: Why Do Poor Sampling Probes Distort Gas Composition?
The sampling probe is the first interface between the process and the gas analyzer system. Therefore, it directly affects sample quality from the beginning. Unfortunately, probe design is often underestimated. If the probe location is incorrect, the extracted gas may not represent actual process conditions. Dead zones, uneven flow distribution, and excessive turbulence can all create biased measurements. At the same time, insufficient filtration allows dust and particulates to enter the system, increasing blockage risk and damaging downstream components.
For harsh industrial environments, reliable probes usually require:
- Corrosion-resistant materials
- High-temperature capability
- Integrated primary filtration
- Heated structures to prevent condensation
Modern heated probes often combine filtration and temperature control into one compact assembly to improve sample stability. Without proper probe design, the sampling system already loses accuracy before transport even begins.
Hidden Problem #3: Why Are Heated Sample Lines So Critical?
Many gas analysis failures happen during sample transport. After extraction, the sample gas may travel several meters or even tens of meters before reaching the gas analyzer. During this journey, temperature management becomes absolutely critical. If the sample line loses heat, several problems appear almost immediately:
- Condensation forms
- Dust accumulates
- Acid corrosion begins
- Response time increases
- Measured values drift
This explains why heated sample lines are considered standard practice in industrial extractive gas analysis systems. Their main function is keeping the gas temperature continuously above the dew point during transport.
Modern heated lines typically include: Electric heat tracing, thermal insulation, PTFE or stainless-steel inner tubing, and temperature sensors and alarms. Some hot-wet extractive systems even maintain temperatures above 180°C to prevent acid condensation. Without proper heating, even a high-end gas analyzer cannot produce stable results.
Hidden Problem #4: Why Does Incorrect Sample Conditioning Create False Readings?
Many engineers focus heavily on filtration while ignoring chemical compatibility. However, improper conditioning materials can quietly change gas composition during transport. For example, some filter materials adsorb target gases onto their surfaces. Certain tubing materials may also react with corrosive compounds over time. Although these changes are often invisible, they directly affect gas analyzer accuracy. That is why industrial sampling systems commonly use chemically inert materials such as PTFE, PFA, and stainless steel. These materials minimize adsorption and maintain sample integrity throughout the transport process.
At the same time, conditioning systems must balance filtration efficiency with response speed. Excessive filtration may improve cleanliness but also slow gas analyzer response time significantly. Therefore, good sample conditioning is never just about “cleaning the gas.” The real goal is preserving the gas without changing its composition.
How Can Engineers Prevent Most Sampling System Failures?
Before designing the gas sampling system, engineers must fully understand the operating environment. Important parameters include: gas temperature, process pressure, moisture level, dust concentration, and corrosive gas content. These factors directly determine probe design, filtration method, heating requirement, and conditioning strategy. Although industrial conditions vary greatly, most sampling problems can be prevented by following several core principles.
First, always maintain the sample temperature above dew point during the entire transport process. This prevents condensation and analyte loss.
Second, use chemically inert materials throughout the sampling path. This minimizes adsorption and chemical reactions.
Third, continuously monitor system temperature, flow, and pressure. Early alarms help operators identify problems before measurements drift.
Finally, design the system for maintenance access. Easy filter replacement and probe cleaning greatly improve long-term reliability.
In practice, stable sampling systems are usually simple systems with strong temperature control and reliable flow management.
Conclusion
Gas sampling system may look simple from the outside, but in reality, it is one of the most technically demanding parts of industrial gas analysis. Every section matters, from probe placement and heated transport to filtration and conditioning. If any stage changes the original gas composition, the gas analyzer no longer measures the real process condition. That is why the best sampling systems focus on one core objective: Deliver the sample gas to the gas analyzer exactly as it exists inside the process.
Once engineers fully understand this principle, gas analyzer accuracy, system reliability, and long-term operational stability improve dramatically. If you need a tailored solution, contact with us please!
FAQs:
Q: What is a gas sampling system in a gas analyzer?
A: A gas sampling system extracts process gas and delivers it to the gas analyzer without changing its composition. It usually includes a sampling probe, filters, heated sample lines, pumps, and conditioning components. Its main purpose is maintaining sample integrity during transport and analysis.
Q: What causes condensation in gas sampling systems?
A: Condensation occurs when sample gas temperature falls below its dew point during transport. This commonly happens in unheated or poorly insulated sample lines. Once condensation forms, water-soluble gases may dissolve into liquid droplets, causing inaccurate readings.
Q: Why are heated sample lines necessary?
A: Heated sample lines keep the gas temperature above the dew point throughout transport. This prevents condensation, protects analyte integrity, and improves measurement reliability. Heated lines are especially important in CEMS, flue gas monitoring, and high-moisture applications.
Q: What temperature should a heated sample line maintain?
A: The sample line temperature should remain higher than the water or acid dew point of the gas stream. Many industrial systems maintain temperatures between 120°C and 180°C, depending on gas composition and application requirements.
Q: What happens if the sample gas cools too quickly?
A: Rapid cooling can cause condensation, adsorption, delayed response time, and corrosion inside the sampling system. As a result, gas analyzer readings become unstable or falsely low. Sensitive gases like SO₂ and H₂S are especially vulnerable to loss during cooling.
Q: Why is the sampling probe so important?
A: The gas sampling probe is the first contact point between the process gas and the gas analyzer system. A properly designed probe improves representativeness, blocks particulates, and prevents condensation near the extraction point. Poor probe placement can lead to biased measurements.
Q: What materials are commonly used in gas sampling systems?
A: Industrial sampling systems often use PTFE, PFA, stainless steel, and other chemically inert materials. These materials resist corrosion and minimize adsorption of target gases during transport.
Q: What is the difference between direct extraction and dilution sampling?
A: Direct extraction transports the sample gas directly to the gas analyzer and is suitable for moderate conditions. Dilution sampling mixes the sample with clean dry gas before transport, making it more suitable for high-temperature or highly corrosive applications like cement kilns and coal-fired boilers.























